Take Back your Time: How Artificial Intelligence Can Support Breast Screening Business Continuity in Your Practice
Nov 26, 2020Categories: Blog, Breast Cancer Risk, Breast Density, Mammography Image Quality, Operational Efficiency
Artificial intelligence continues to be one of the hottest topics in radiology, promising transformative solutions for breast cancer screening. However, making sense of how A.I. fits into your current breast screening workflows can feel like a complex undertaking. At Densitas we have committed to help you navigate through the discourse on how A.I. can assist you and augment your mammography practice, providing actionable insights for better reporting workflows and improved operational efficiencies in mammography. In a time where COVID-19 has created a growing backlog of delayed screening mammography services, clinical care teams are experiencing burnout and are looking for workflow efficiencies to manage their increasing workload.
To help you visualize A.I. in your breast screening practice, we have identified some of the key challenges that radiologists and clinical care teams experience, and how A.I. is poised to respond.
Take Back your Time with Stress-Free Audits for MQSA EQUIP Compliance

Breast imaging facilities must be certified through the Mammography Quality Standards Act (MQSA), requiring annual FDA MQSA EQUIP audits and accreditation renewals. The United States sees over 40 million mammograms performed annually, across nearly 9,000 accredited mammography facilities. Operating at such high capacity, and with looming projections of even greater increases in volume, it is an overwhelming feat for mammography facilities to maintain MQSA EQUIP compliance.
MQSA guidelines are in place to ensure mammography facilities consistently deliver high-quality patient care. The main challenge that facilities face is the administrative burden associated with the establishment of the required quality procedures and demonstrating they are following those procedures.
The majority of centers are still using time-consuming paper-based documentation of quality assurance processes. Clinical care teams shoulder the administrative burden of adhering to these resource-intensive requirements for accreditation image selection, image quality assessment, technologist reviews, management of documentation of corrective actions, and EQUIP audit report compilation.
Incorporating the newest A.I. solutions into your mammography enterprise can enable evidence-based care delivery with time-efficient and cost-effective continuous quality control in compliance with FDA MQSA EQUIP, and on-demand audit report generation so that more time is spent focusing on patient care.
Eliminate Costly Technical Recalles using A.I. Automation for Image Quality Assessment Before your Patient Leaves

Accurate patient positioning in mammograms is sometimes difficult to achieve, accounting for the majority of inadequate quality mammograms, and results in the sensitivity of mammography dropping from 84% to 66%.
In standard mammography practice, at the time of image acquisition, technologists do not have a second reader assessment of clinical image quality to rely on, and positioning errors are typically identified post-study, which triggers technical recalls. Technical recalls are costly to the healthcare system in many ways: they are not reimbursable, they add to the growing COVID-induced backlog of screening mammography exams, and increase psychological distress in patients.
Groundbreaking A.I. technologies have opened the door to automated clinical image quality assessments at point-of-care allowing repeats before a patient leaves the exam room and eliminates the need for costly technical recalls. This is important now more than ever with mammography facilities extending their hours of operation to mitigate the negative impact of COVID-19 on breast screening services, and as mammography exam visits have become longer to adhere to both PPE and social distancing good practices.
Rapid Short-Term Risk Scores Inform Development of Tailored Follow-up Screening Protocols

Most traditional lifetime breast cancer risk models require extensive and tedious collection of clinical patient history and other unreliable risk factor data that can be subject to recall bias, and can often be unavailable. Long-term risk models tend to overestimate cancer risk and perform increasingly poorly the farther out the projected time.
The emergence of electronic systems for collection of risk factor data does little to overcome these challenges, and obtaining routinely stored data from disparate data sources is complex and often infeasible due to lack of interoperability between information systems. Even if the information systems allow on-demand availability of data access, it is time-consuming for healthcare systems to collect all the necessary risk factor data.
Breast cancer risk models for use in screening mammography at a population level can be simplified and made more practical through the use of a smaller set of readily available clinical risk factors derived directly from the mammogram. Risk estimates from these models are reliably reproducible, available on-demand at point-of-care, and provide actionable information that can be used to develop tailored follow-up screening protocols and better inform reading assignment of studies to designated mammographers.
By using standardized risk factors from DICOM images, a short-term rapid risk assessment can be used to reliably and reproducibly evaluate breast cancer risk for every woman who has a mammogram such that the risk estimates are comparable and available across the entire population, and provide actionable information at point-of-care for immediate clinical decision-making.
Standardize Breast Density Assessments for Consistent Stratification of Patients for Precision Breast Health
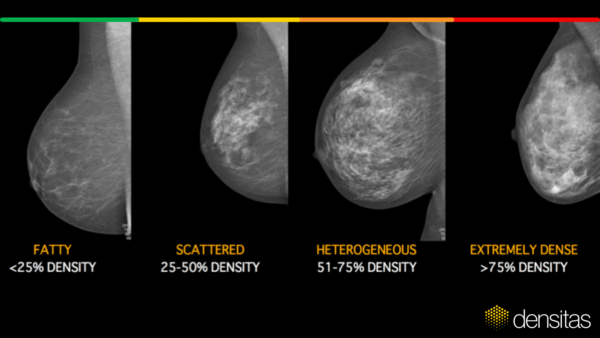
Breast density is an independent risk factor for breast cancer and standardized breast density assessments are crucial to ensure reliable risk estimates. Visual breast density assessments by radiologists have been shown to be highly variable.
The evolution of breast density algorithms has spanned nearly twenty-five years from the mid-1990’s, starting with pixel thresholding methods applied to digitized films, followed by volumetric algorithms based on physics models applied to RAW digital images, and culminating today with machine learning and deep learning algorithms that are trained on massively large data sets of standard DICOM digital images.
Volumetric breast density algorithms first made their appearance in 2008 and are based on physics models that generate quantitative breast density measures that are thresholded at ad-hoc cut-points to define a four-category breast density scale. However, in 2013 the BI-RADS Atlas 5th ed. density scale replaced the older 4th ed. scale and was a significant move away from a quantitative scale to a qualitative four-category breast density scale based on the appearance of breast tissue on a mammogram.
While volumetric density algorithms were unable to evolve to address the fundamental shift in the ACR BI-RADS Atlas breast density scale guidance, modern deep learning algorithms were particularly suited to accommodating the new ACR BI-RADS Atlas 5th ed. breast density guidance, providing breast density assessments aligned with radiologists’ visual assessments of breast density and making it available at the radiologist’s fingertips at the time of image acquisition and reporting.
Automated breast density assessments from deep learning A.I. algorithms provide a practical way to implement standardized on-demand risk-based stratification of patients across entire health systems for worklist prioritization and compliance with breast density reporting requirements.
Set up a meeting now with our team to discover how A.I. can improve your mammography practice.
Let’s Stay Connected, Subscribe for Updates
Join our email list to stay up to date on the latest advancements in breast health technology.
 Canada
Canada USA
USA UK
UK